In addition, the proliferative capacity of these NCC is also diminished. 
Although, Pax3 heterozygous mice do not show ENS defects, compound Pax3 Tcof1 heterozygous mice exhibit cumulative apoptosis which severely reduces the NCC population that migrates into the foregut. In contrast, Tcof1 haploinsufficiency is able to sensitize Pax3+/- mice to colonic aganglionosis. This alone, however, is not sufficient to cause colonic aganglionosis as alterations in the balance of NCC proliferation and differentiation ensures NCC colonize the entire length of the gut of Tcof1+/- mice by E18.5. Haploinsufficiency of Tcof1 in mice results in a reduction of vagal NCCs and their delayed migration along the length of the gut during early development. We have discovered that Tcof1 may be one such modifier. 
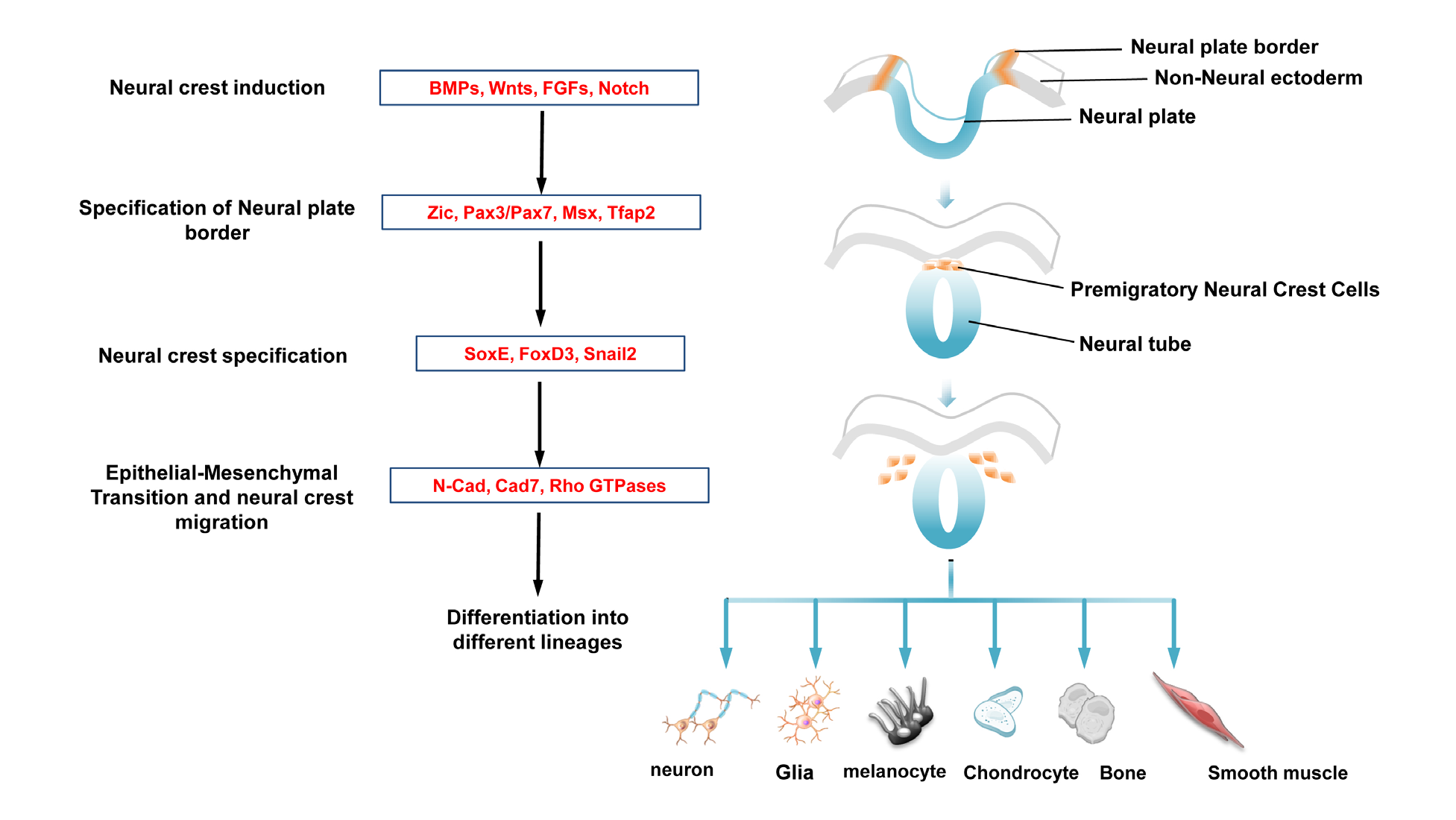
Therefore, additional unidentified genes or modifiers must contribute to the etiology and pathogenesis of Hirschsprung disease. Although extensive research has identified many key players in the pathogenesis of Hirschsprung disease, a large number of cases remain genetically undefined. The inheritance of HSCR is complex, often non-Mendelian and characterized by variable penetrance. These ganglia comprise the enteric nervous system (ENS) and are derived from migratory neural crest cells (NCCs). Hirschsprung disease (HSCR) is a human congenital disorder, defined by the absence of ganglia from variable lengths of the colon.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
